Imagine for a moment, that instead of bacteria being your enemy, they can become your weapon against disease? That by manipulating their good traits, we can use them to work for us? It is true that often we see bacteria in a negative light, as sources of infection and disease, but beneath this bad reputation is a real potential for solutions to many of our human problems.
Bacteria – A Brief History of Time
Bacteria are the most diverse and abundant life forms in the world, with over 70,000 known species. They survive and thrive in every environment and location – be it in icy glaciers, fiery volcanoes or even within the acidic stomach. While we have all heard of them, how much about their function and structure do we understand, even now?
Well, their cells are structured in a relatively simple fashion – they break down nutrients, chemicals or light to provide them with energy, and they then must remove or excrete the waste produced; essentially much the same as us humans, albeit in a simpler system. It is due to this principle of a simple system, that bacteria have evolved or developed many different adaptations to help them survive better in particular environments. Already, humans have observed some of these bacterial adaptations, and have seen the benefits and applications – such as using the ability of Lactic acid bacteria to produce acid in milk, leading to cheese and yogurt production, as illustrated in figure 1.
Often, we now see that many environmental or health issues we humans deal with, can benefit from bacterial intervention. Using antibiotics and bacteria-produced vaccines has helped defeat many illnesses, and perhaps this role can be expanded even further by exploring the bacterial world. Using or modifying bacteria for benefits has long been a staple of research, and can perhaps be misinterpreted by the public due to the controversy surrounding the phrase “gentically modified”. However, for research purposes, this is a saft and long established practise, allowing us to utilise bacteria for increased benefit. This method was first truly used to allow insulin, for diabetics, to be produced on mass, using genetically modified E.coli cells.
The Miniature Factory
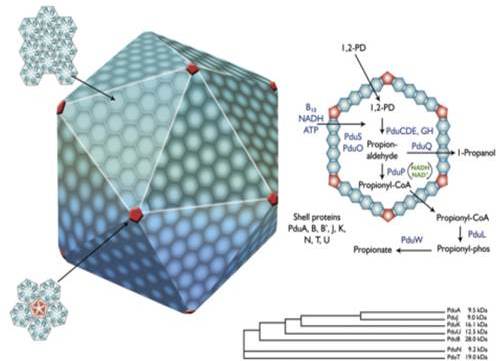
Approximately 20% of known bacteria contain a small, distinct structure within their cells, called a bacterial microcompartment. These microcompartments, or MCPs, consist of a hollow shell, which is made completely of protein. Their hollow interiors provide a space for various chemical reactions to occur, protected from other parts of the cell.
This ability to produce an MCP appears to play a role in the survival and capabilities of the bacterial cell – after all, bacteria are a relatively simple system, so why would they invest several percent of their genes in making these MCPs unless they offered a benefit for them? Waste not, want not is a key motto of the bacterial world.
For example, imagine that these MCPs are like little factories, inside the bacterial cell. They offer an enclosed space where reactions or processing can take place. Like a factory, they take in a particular material, and produce a product of benefit. This must be an efficient and speedy process, which must not unduly waste the effort and energy of the cell and so is only turned on when the raw materials are available to make the product. To avoid disruption to other essential chemical reactions going on at the same time, the factory walls keep the work process separate from the rest of the cell. Having all the specialized machinery (enzymes) in the same place makes the whole process more efficient.
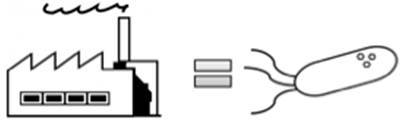
In this role, the MCPs work to selectively take in chemicals originating from the cell’s metabolic processes or from the cell’s surroundings, and then allow a process or reaction to occur within the space of the MCP. These reactions can be diverse, perhaps removing metabolic waste from the cell, or breaking down a chemical to allow it to be used by the bacteria.
Differences & Associations
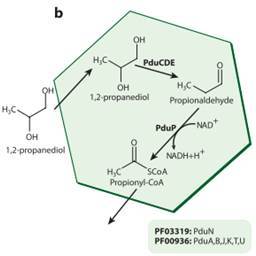
There are an increasing number of different types of MCP now recognised, in many different species of bacteria. The Pdu MCP, which is the focus of my research, is activated by the presence of the chemical 1,2-propanediol (1,2-PD) in the environment around the bacterium. The bacterium can then take this 1,2-PD, and start chemical reactions in the Pdu MCP, resulting in the production of a new chemical, propionaldehyde, which it can process into two more chemicals which the bacterium can use for energy and food. The Pdu microcompartment can also transform a slightly different chemical, glycerol, to an antibacterial chemical, 3-hydroxypropionaldehyde, also known as reuterin, and even further transform reuterin to 1,3 propanediol which is used as a building block in the production of polymers (large molecules).
The Eut MCP is another common type of MCP, which takes up the chemical ethanolamine. This Eut MCP is associated with nasty or pathogenic strains of E.coli, and Salmonella bacteria which cause serious food poisoning. In the right conditions in the intestine, the Eut MCP helps these bacteria to grow much faster than their competitors and cause disease.
But how can this process, or these MCPs play a role in our lives? Well, using molecular technology and by genetically altering, or modifying the bacteria, we can manipulate and change different MCPs. In the cases of good bacteria, we can use the MCPs to process specific chemicals, leading to the production of useful products. Or, in the cases of bad or pathogenic bacteria, we can try to use metabolic processes in the MCP to damage or destroy the bacterium which contains the MCP, which may prevent the bad bacteria from causing infection.
Study Aims
As part of my research, I focus on bacterial MCPs, particularly in the probiotic species Lactobacillus reuteri. My research is based around the idea or hypothesis of using the metabolic activity of bacterial MCPs for disease therapy options. In particular, I am using the case study of hyperphosphatemia, a medical condition where excess phosphate builds up in the bloodstream in patients with kidney failure due to excessive phosphate absorption from the gut. This can cause many health problems, with the only current resolution involving expensive pharmaceutical medication.
If we could use safe MCP-containing bacteria to take in the excess phosphate from the gut, and remove it even temporarily by a reaction in the MCP, could we possibly use this as a therapy? This concept has been coined by our laboratory as the Microcompartment Enhanced Biological Phosphorus Removal (MEBPR) – the expression of a phosphate storing enzyme (ppk) within a MCP inside bacteria.
Answering this question is my primary research focus, and requires using several different methods. Lactobacillus reuteri has many desirable traits as a bacterial model, which is why I use it for my research. It has a naturally occurring pdu MCP, which I previously mentioned, and which is triggered by the presence of the chemical 1,2-propanediol. Also, a key aspect of using this strain is its status as a GRAS (generally regarded as safe) probiotic bacteria, which means it is very safe for metabolic rewiring.
Lactobacillus reuteri also has a natural phosphate storing enzyme. My objectives include the possibility of using this enzyme to store excess phosphate from the environment, using the MCP as a focal point for these reactions to occur.
At this current stage, many of the enzyme-MCP constructs have been created, and will soon be tested in the laboratory. Within the bacteria, increased phosphate storage can already be seen, but we don’t yet know if this will be enough to make the bacteria effective phosphate removal machines.
Conclusion
If this experiment is successful in its objectives, the ability to use MCPs in probiotic bacteria would open up many possibilities for therapeutic use. Whilst there are still many obstacles regarding this step, it could become a hugely beneficial and successful tool for treating certain illnesses and ailments.
Microorganisms, and bacteria in particular, hold the secrets to many things due to their ancient age on this planet. Already we exploit and use them to enhance our lives, through food fermentation, the product of alcohol and even the production of antibiotics – could it be possible to add the exploitation of bacterial microcompartments to this list?
Research, exploration and creative thinking may reveal more.
Karen McCarthy is a Ph.D. student in the Department of Microbiology and Alimentary Pharmabiotic Centre, under the supervision and support of Prof. Mike Prentice. She is funded by the Health Research Board (HRB).

