Introduction
Fungi are one of the major health concerns in modern life. It is known that up to 20% of patients with blood stream infections in intensive care units are affected by disease producing fungi such as Candida and Aspergillus, sometimes dominating the infections in doses that could be lethal. Patients who are immune-compromised/ immune-suppressed – including the elderly, HIV-infected patients, chemotherapy recipients, and transplant patients - are more prone to fungal infections. There are anti-fungal drugs available, but they are expensive and can have severe side effects such as nephrotoxicity (kidney damage). In addition, a major concern is that fungi such as Candida can become drug-resistant. Therefore there is a pressing need to identify new drugs to treat fungi and the diseases associated with them.
- chemical- based drug discovery has not worked
- virtually all the drugs are from microbial origin
- There is a need to explore new environments to avoid rediscovering the same classes of drugs.
Life saver – The sponges
Oceans cover about 70% of the earth and it is highly diverse in terms of its wealth – the marine organisms. It has been seen previously that microbes, such as bacteria and fungi, produce natural substances that can have applications in the pharmaceutical industry, and there are likely to be many undiscovered compounds produced by trillions of microbes present in the world that could have major positive health implications. In our work, I am focusing on microbes that are associated with marine organisms called sponges (Figure 1), because these are known sources of bioactive compounds (substances, like antibiotics, that have an effect on the living organism). One such example of a bioactive from sponges is Jasplakinolide, which is anti-cancer drug.

Sponges are aquatic animals; the great majority are salt water species, living in habitats ranging from the tidal zone to
the deep sea, and around the world from the Polar Regions to the tropics. They are usually found attached to rocks but some
get attached to soft areas. They are simple animals and
do not have any organ systems. Sponges are immobile filter-feeders, acting like chimneys as they pump large volumes of water
through their system and filtering out what they need to eat, and pushing out what they don’t. Microbes such as bacteria and
fungi that are pumped in with the water can take up residence in the sponge. Sponges need to have a strong community of “defence
microbes” because of their tremendous exposure to pathogenic microbes. The area that is being targeted for research is the
bioactive compounds that these microbes produce as part of their natural chemical defence system.
Methodology
For my research, live sponges are first collected and then I look to see if the microbes living inside them produce any interesting anti-fungal compounds. The sponge which I am working with is called Haliclona simulans. Once the preliminary screening is done, the bacteria associated with the sponge are grown in liquid media for few days until they produce the active compound (Figure2).
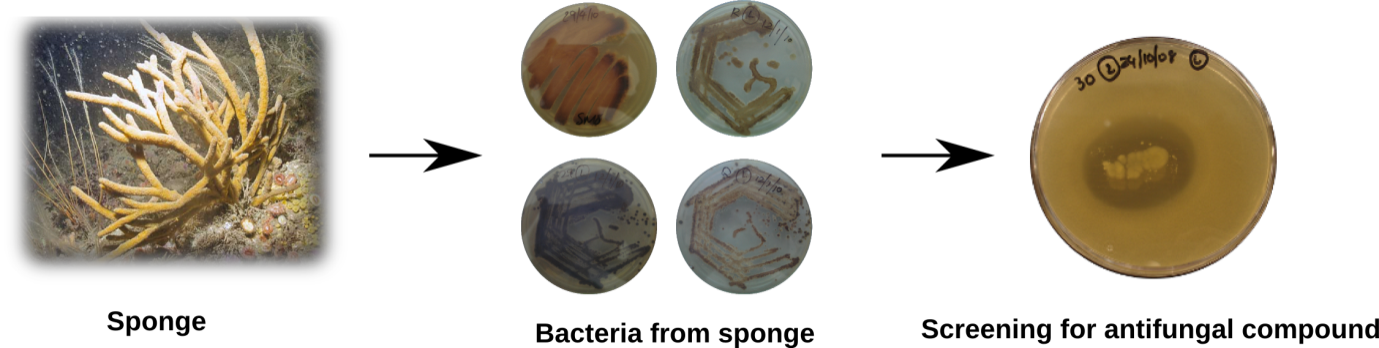
At this stage, the anti-fungal compound is extracted from the bacteria with organic solvents such as methanol, and then purified
using a separation technique called HPLC (High-Performance Liquid Chromatography).
Results
In our laboratory, I have recently extracted, separated and identified a new compound from sponge-associated microbes that has anti-fungal activities (Figure 3). The purified compound was analysed by LC-MS (Liquid Chromatography - Mass Spectrophotometer), a common method for identifying known compounds by determining its mass. The unique mass of our isolated compound indicates that it is a new discovery. The next step will be to obtain the chemical structure of the compound using NMR (Nuclear Magnetic Resonance) spectroscopy. This will help us to identify how the compound behaves both chemically and biologically.
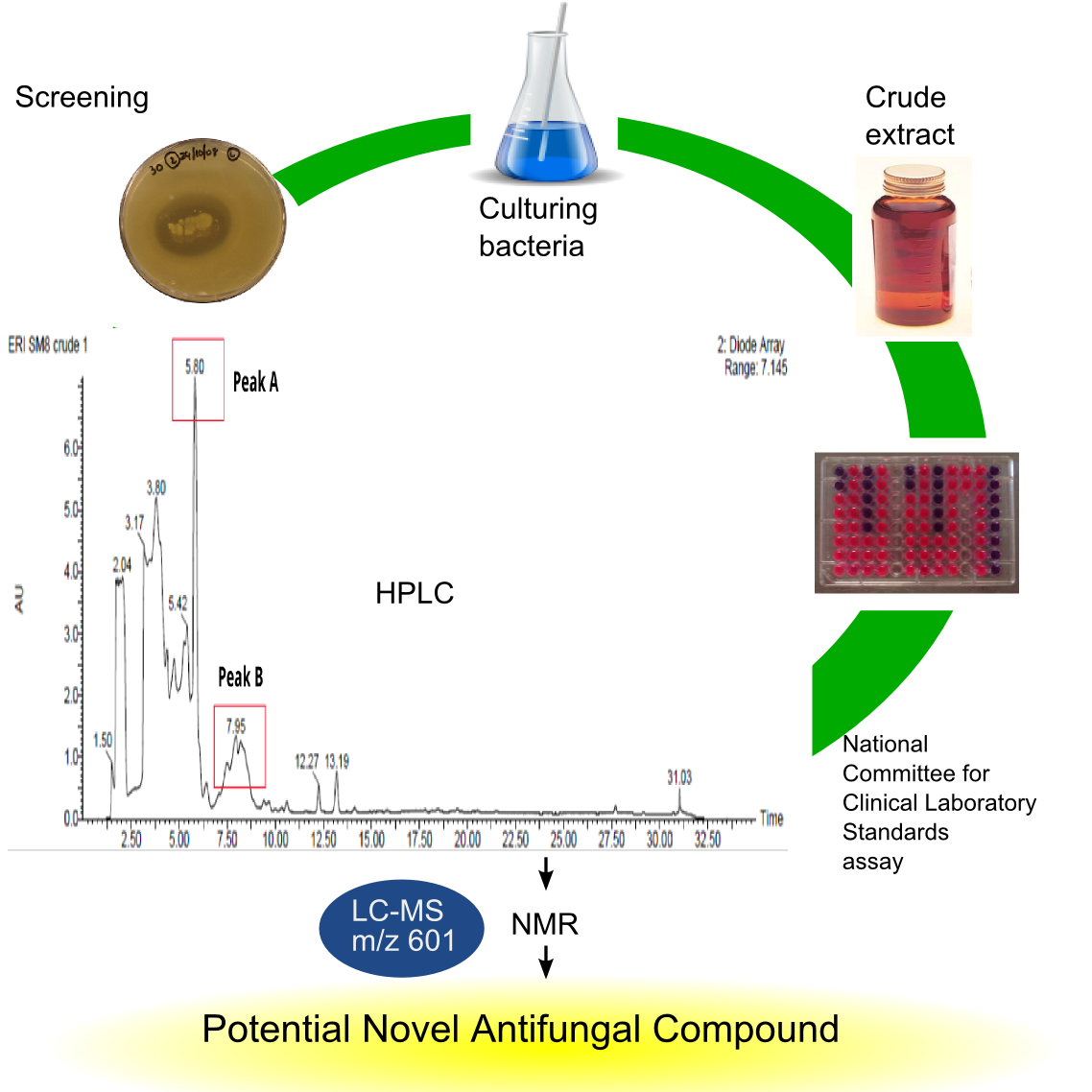
Prospects
Although there is some way to go, this work with sponge-associated microbes and the compounds that they produce could already offer hope to immune-compromised patients who are suffering from secondary, potentially deadly, fungal infections. Further work will involve testing whether these compounds have therapeutic potential, and expanding my screens to look deeper into the sponges present in our oceans for other life-saving drugs.
I would like to thank my supervisors Dr.John Morrissery, Prof. Alan Dobson and Dr.Jonathan Kennedy. I would like to acknowledge the Beaufort Bio discovery Awards Project for funding my research. I would also take this opportunity to thank Dr. Florence McCarthy at the ABCRF in UCC who has been collaborating with us for the LC-MS analysis. I would like to thank Dr. Sarah Knight from NUIG for helping me in the preparation of the article.

